
From early design through to operational readiness, Exyte delivers at every stage.
With over 20 years of experience bringing complex facilities to life, we’re proud to support the biopharma and biotech industries, helping improve health and quality of life for millions around the world.
Our ability to ensure best-in-class workflows, full regulatory compliance, flexible modular layouts, and cutting-edge cleanroom solutions into every project, is what truly sets us apart.
We focus on doing things right: delivering on time, staying on budget, and always meeting the highest technical standards.

Pharmaplan is now Exyte
With the acquisition of Pharmaplan, our service offering now combines decades of process engineering and GMP expertise with the full force of Exyte’s global engineering and execution power.
As pharma and biotech companies harness the power of AI, digital twins, and data-driven discovery, we’re accelerating facility delivery with the same bold vision—integrating smart engineering, modular construction, and sustainability from day one.
Because every second counts in the race to save lives — and every life deserves the best science has to offer.

EPC and EPCM service management
At Exyte, we know every biopharma facility is unique, which is why we offer both EPC and EPCM delivery models. Our EPC approach provides a fully integrated solution, with Exyte taking complete responsibility for design, procurement, and construction - delivering speed, cost certainty, and reduced client burden, especially for complex projects or tight timelines.
For clients seeking greater involvement and flexibility, our EPCM model ensures transparent collaboration, with Exyte overseeing design and construction management while clients retain control over procurement and contractor selection. Backed by decades of life sciences expertise, Exyte adapts seamlessly to each client’s needs, ensuring facilities are delivered efficiently, reliably, and to the highest standards of quality.
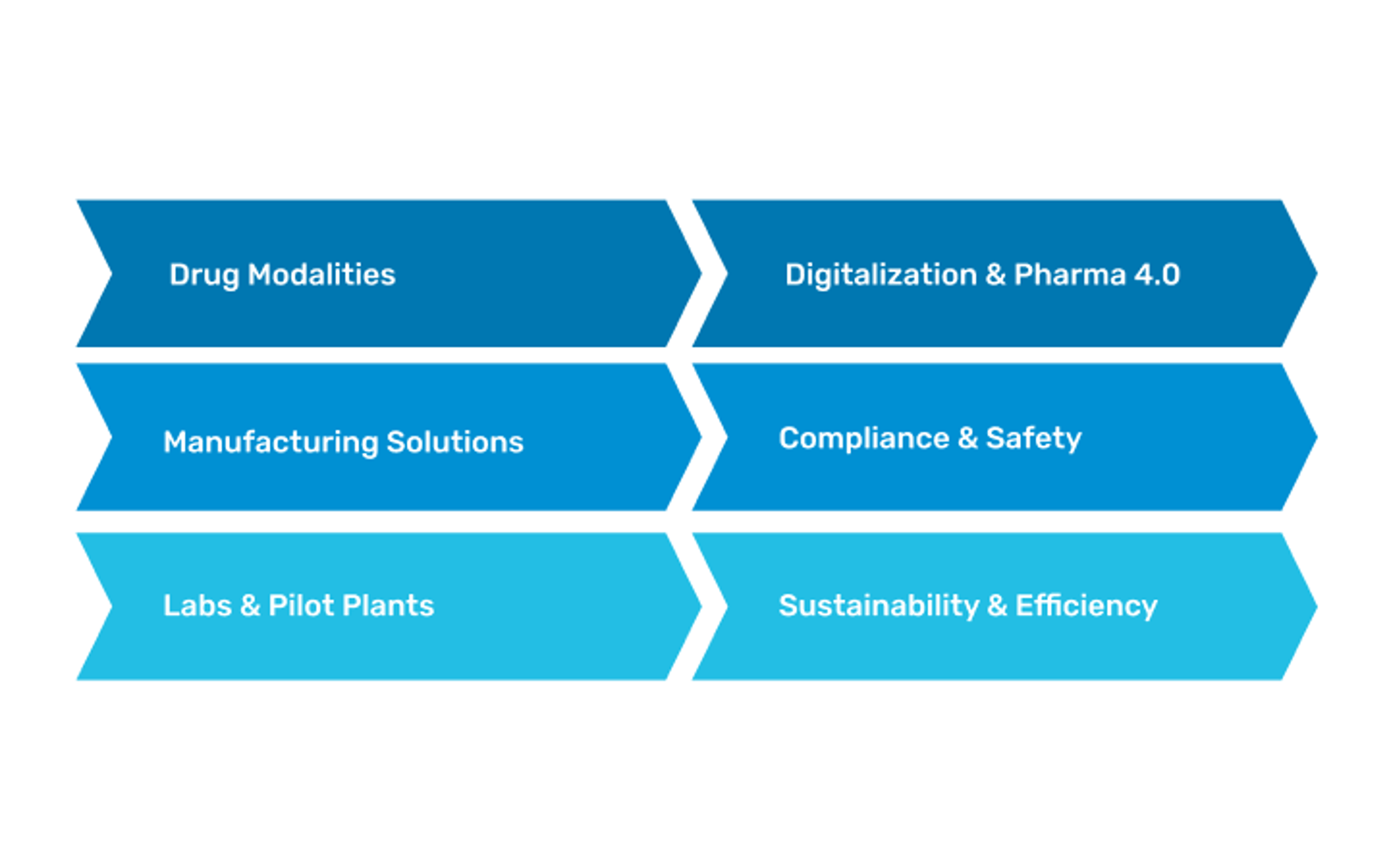
Pharma facility solutions
We deliver end-to-end solutions for pharmaceutical facilities - from R&D to large-scale manufacturing - enabling clients to bring innovative therapies to market faster.
Our expertise includes:
- Drug modalities: Biologics, ATMPs (cell & gene therapies, xRNA, viral vectors), small molecules, vaccines, radiopharmaceuticals
- Manufacturing solutions: Aseptic fill & finish, oral solid dosage, continuous manufacturing
- Laboratories & pilot plants: R&D, QC, MSAT, preclinical testing
- Digitalization & Pharma 4.0: Automation, MES, digital twins, data-driven operations
- Compliance & safety: Global cGMP expertise, CQV, CSV, biosafety, EHS
- Sustainability & efficiency: Green facilities, energy recovery, modular concepts

Interactive virtual immersion
Cutting edge technology is weaved across all areas of our offering, including the early design stage.
With a range of digital twins and AI tools that allow real-time facility visualization, Virtual Campus is our newest immersive experience, allowing you to identify issues early and train operators, all before construction begins.

Experts and innovations redefining excellence at Exyte
At Exyte, innovation in biopharma and life sciences is driven by more than engineering excellence – it’s powered by our people. Our team of Subject Matter Experts spearhead innovation, bringing decades of hands-on experience, regulatory insight, and future-ready thinking to every project. From cutting-edge biopharma facilities to compliant, scalable solutions, their expertise shapes the industry’s future through the vision, leadership, and passion that fuels Exyte's position at the forefront.